Introduction
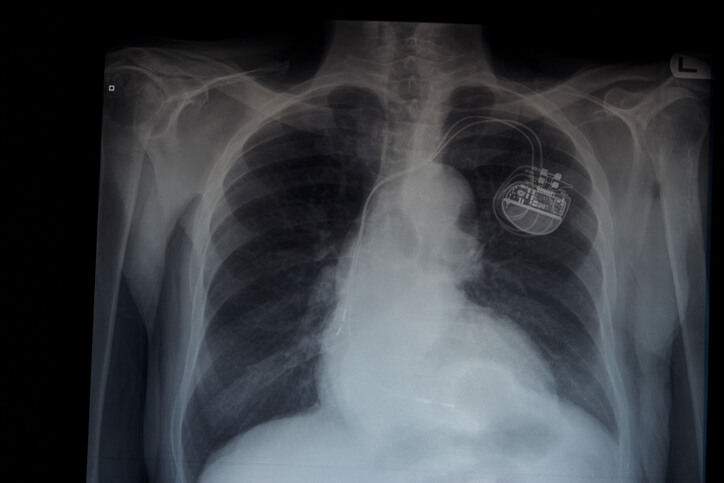
The Medtech industry is booming with the invention of its revolutionary health care products. One such product is pacemaker that is used in patients with irregular heartbeats. Arrhythmias and bradycardia patients are hugely benefited with this product as these products are reliable regarding irregular heart beat issues. Medtronic is an Irish company famous for its world-class health technology, and solutions like Micra, the world’s smallest pacemaker. This pacemaker is less invasive as it is placed directly into the heart, thus the chances of chest incision, scar or burn have significantly come to zero. Micra is 93% smaller when compared to other conventional pacemakers. Its size is similar to the scale of any large vitamin capsule.
Dr. Bruce Lindsay, who is the senior most doctors at the Cleveland Clinic’s Heart and Vascular Institute and section head of Cardiac Electrophysiology and Pacing department, stated that the medical institutes across the globe, including Cleveland Clinic are being pressured to keep the costs down for such devices.
Key applications of Micra: World’s smallest pacemaker
With Micra, various health care professionals get the opportunity to study bradycardia and Arrhythmias disease. Medtronic helps such individuals in organizing educational workshops, where the needs of each group are catered. This includes:
- Referring and Implanting Doctors.
- Allied Medical Professionals.
- Electrophysiology and Cardiology Associates.
- Hospitals staff and Administrators.
These professionals can also take help from Micra TPSTM Academy, where they can get support and further education training related to Micra Transcatheter Pacing System(TPS).
| Recommended for you | |
| A wearable MRI | |
| 13 themes of preventive medicine | |
| Digital prevention needs dialogue between doctors and IT professionals |
Salient technical specification
The world’s smallest Pacemaker is gaining popularity due to its incredible features:
- It has 12 years of battery longevity.
- MRI Sure Scan features lets you scan by using 1.5T or 3T full body MRI.
- Rate Response is determined using Accelerometer.
- Compatible with CareLink 2090 Programmer. Thus, accessories are not required.
- Assistance with Complete Capture Management.
- Ultra-low-power circuit helps in longer battery life that is up to 12 years.
Who are the target user groups?
Micra can be used considering the age and medical conditions of the patients. Till now, there are no complications while using this device. Following groups can use this pacing device to control their heart rate.
- Permanent high-grade AV block which is referred as Symptomatic paroxysmal in the presence of AF, wherein dual chamber pacing with lead is involved.
- Permanent high-grade AV block or Symptomatic paroxysmal in the absence of AF wherein dual chamber pacing with lead is required.
- Sinus node dysfunction, which includes sinus pause/sinus bradycardia or Symptomatic bradycardia-tachycardia syndrome.
Following patients are not advised to use Micra:
- Patients who are intolerant to heparin.
- Having difficulty in tolerating 1.0 mg dexamethasone.
- Having difficulty in accommodating a 7.8 mm introduced on the right side of the heart.
- Issues with a mechanical tricuspid valve.
Outstanding market of leadless pacemaker: Micra
Micra- the pacemaker can be easily implanted through endoscopy without the need of conventional surgery. This has significantly reduced the costs of operation, and hence standard lead pack and hence thereby enhanced the process of implantation. The Journal of the American Medical Association and FierceMedTech have recently stated the advantages of leadless devices on their website. This contributes to the positive growth perspective of the Micra. The outlook for Medtronic’s cardiovascular group has suggested that there is substantial growth potential to Micra due to its adoption of lead-free technique. This will result in declination of cardiac rhythm management market share further stabilizing the Medtronic company.
As the per Berkley’s report, the normal rate of implantation is $14,920, depending on the complication costs that are involved in a pacemaker process, varying between 0% to 16% and having the average value of 5%.
Exciting plans and competitors
To reimburse the development costs of Micra which is estimated around $100 million, electronics could charge more in excess crossing $10,000 per device.
To increase sale and gain customers, the company is likely to position Micra very competitive as compared with traditional pacemakers, depending on a total cost basis. This is the reason that company is planning to sell Micra TPS at low cost in emerging markets like India.
The FDA approved Micra TPS is having fierce competition with Nanostim which is leadless pacemaker device from a St. Jude Medical. Nanostim had won CE Mark back in the year 2013 and further waiting for its FDA approval from the US.Dr.Lindsay believes that the price of Micra may decline as their competitors are entering emerging markets like US and India.
Conclusion
TPS which is the Micra Transcatheter pacing system is inserted directly into the right ventricle without any use of leads. The cost of this product is estimated to be around $10,000 which is pretty high compared to conventional pacemakers with an average price of $2,500. The promising features of this leadless pacemaker give lots of hope in treating the heartbeat issues of various cardiac patients across the globe.
Image credit: www.istockphoto.com